Uncommon-earth metals are indispensable for a lot of technical merchandise, from smartphones, laptops, batteries, electromotors, and wind generators, to catalysts. A Japanese crew has now launched a molecular “cage” with “caps” that can be utilized to restrict sure rare-earth-metal ions selectively for isolation or recycling.
The rare-earth parts embrace 17 metals: scandium, yttrium, lanthanum, and the lanthanides, the 14 parts that observe after lanthanum within the periodic desk, together with neodymium and europium. The identify is deceptive as a result of the rare-earth metals should not really uncommon. They’re all over the place within the setting however are extremely dispersed and sure in minerals (“earths”); giant deposits are uncommon.
Reclaiming these parts from digital waste is changing into extra essential. Some microorganisms have been found that include enzymes with rare-earth metals. These might be helpful in extraction and reclamation and supply inspiration for the usage of rare-earth metals as catalysts.
Uncommon-earth-metal ions are additionally present in our bodies of water and in effluent. Nonetheless, they’re onerous to separate individually from aqueous options. One motive for that is that they’re often hydrated, which means that they’re sure to water molecules. Their states of hydration are completely different and will change. This makes identification and isolation of the ions via binding to ligands harder.
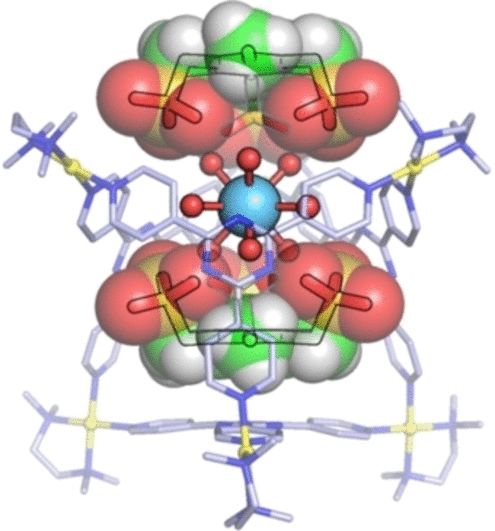
A crew led by Makoto Fujita on the College of Tokyo and the Institute for Molecular Science has now managed to restrict the hydrated types of trivalent ions of a collection of rare-earth metals in closed cages. Every cage molecule consists of 4 natural ligands formed like triangular plates which might be related by their tricks to six palladium ions to make an octahedral cage with two giant openings. The rare-earth-metal ion matches into the cage with its 9 sure water molecules.
Tabuchi et al.
The vital function of the cage is the 2 “caps” that cowl the openings. These are planar molecules with three negatively charged binding arms that bind to the rare-earth-metal ion’s water molecules via hydrogen bridges. As well as, they’re held tight by electrostatic interactions with the positively charged palladium ions within the cage.
Not all rare-earth-metal ions are captured equally properly by this technique. Delicate variations of their radii and most well-liked modes of hydration decide how properly they match into the cages: lanthanum and the early lanthanides, equivalent to europium, are sure considerably extra strongly than the later lanthanides, like ytterbium. Scandium, for instance, solely has six water molecules sure to it and can’t discover a steady place inside the cage. It’s thus barely held in place.
Confinement of hydrophilic metallic species in a closed cavity might be an method for the isolation of rare-earth metals, in addition to for the event of novel catalysts analogous to metal-containing enzymes (metallozymes) in microorganisms.
Assets
-
R. Tabuchi, H. Takezawa, M. Fujita (2022) “Selective Confinement of Uncommon-Earth-Metallic Hydrates by a Capped Metallo-Cage below Aqueous Situations”, Angew. Chem. Int. Ed. doi: 10.1002/anie.202208866