A world crew led by scientists from the Institute for Superconducting and Digital Supplies on the College of Wollongong in Australia has verified that the introduction of novel molecular orbital interactions can enhance the structural stability of cathode supplies for lithium-ion batteries.
In open-access analysis revealed in Angewandte Chemie, first writer Dr Gemeng Liang, Prof Zaiping Guo, A/Prof Wei Kong Pang and associates, used a number of capabilities at Australia’s Nuclear Science and Expertise Group, (ANSTO) and different methods to offer proof that doping a promising cathode materials—spinel LiNi0.5Mn1.5O4 (LNMO)—with germanium considerably strengthens the 4s-2p orbital interplay between oxygen and steel cations.
The 4s-2p orbital is comparatively unusual, however we discovered a compound within the literature by which germanium has a valence state of + 3, enabling an electron configuration ([Ar] 3d104s1) by which 4s transition steel orbital electrons can be found to work together with unpaired electrons within the oxygen 2p orbital, producing the hybrid 4s-2p orbital.
—Dr Gemeng Liang
The 4s-2p orbital creates structural stability within the LNMO materials, as decided utilizing synchrotron and neutron experiments at ANSTO’s Australian Synchrotron and the Australian Middle for Neutron Scattering, in addition to different strategies.
The crew used neutron and (lab-based) X-ray powder diffraction, in addition to microscopy, to substantiate the situation of the doped germanium on the 16c and 16d crystallographic websites of the LNMO construction with Fd3m area group symmetry.
Because the valence state of the germanium dopants was vital to research, laboratory X-ray photoelectron spectroscopy (XPS) and X-ray absorption spectroscopy (XAS) measurements on the Australian Synchrotron had been carried out. They confirmed that germanium dopants have a median valence state of +3.56, with germanium on the 16c and 16d websites being +3 and +4, respectively. The outcomes of density practical concept (DFT) calculations supported this remark.
The researchers evaluated the electrochemical efficiency of batteries containing LNMO and in contrast that with these containing LNMO with 4s-2p orbital hybridization (referred to as 4s-LNMO). These assessments discovered that doping with 2% germanium contributed to superior structural stability, in addition to lowered battery voltage polarisation, improved vitality density, and excessive voltage output.
We wished to grasp the lithium diffusion kinetics within the two supplies and located that after the germanium is launched into the system, the diffusion of lithium within the materials is quicker, permitting sooner cost functionality.
—Dr Liang
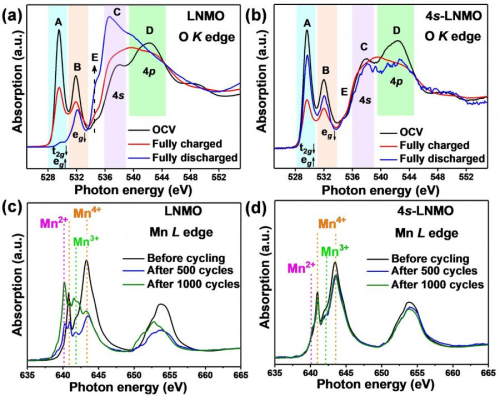
O Ok edge NEXAFS spectra of a) LNMO and b) 4s-LNMO at OCV, fully-charged and fully-discharged states; Mn L edge NEXAFS spectra of c) LNMO and d) 4s-LNMO earlier than biking and after 500 and 1000 cycles.
Following the efficiency testing, Dr Liang used synchrotron-based Close to-Edge X-ray Absorption Spectroscopy (NEXAFS) on the Tender X-ray beamline for extra detailed details about the digital constructions of lively supplies throughout biking. Spectroscopic information on the open circuit voltage of the battery discovered a big enhance within the depth of peaks of the 4s-LNMO materials on the place akin to the 4s-2p orbital hybridization—offering additional validation of the profitable introduction of the novel 4s-2p orbital interplay.
As a result of we will see the unfilled orbitals, these are linked in a definite however sophisticated strategy to the crammed orbitals, we will use these to assist characterise higher the chemistry of the system both by quantum mechanical calculations or by comparability to related supplies.
—co-author Instrument scientist Dr Bruce Cowie
The NEXAFS information had been additionally helpful in evaluating the conduct of manganese within the materials.
We all know that conserving manganese from dissolving into the electrolyte and inhibiting the formation of manganese +2 and +3 within the construction will assist forestall structural degradation.
—Dr Liang
The NEXAFS outcomes confirmed that there was solely a minor quantity of Mn3+ and no noticeable Mn2+ within the 4s-LNMO, which additional will increase the structural stability of the fabric.
In operando experiments on the Powder Diffraction beamline on the Australian Synchrotron explored the structural conduct of the fabric inside a battery throughout biking. Utilizing these information, the crew confirmed the suppression of an unfavorable two-phase response at excessive working voltage within the 4s-LNMO.
Orbital hybridization is sort of a brand new idea in battery analysis, however may be very promising for fixing battery efficiency points. Even higher—this method is extendable to different battery supplies.
—Dr Liang
Different ANSTO co-authors included Dr Anita D’Angelo, Dr Bernt Johannessen, Dr Lars Thomsen, and Prof Vanessa Peterson. Collaborating establishments included The College of Adelaide, the College of Surrey (UK), and the Industrial Expertise Analysis Institute (Taiwan).
Dr Liang, who at the moment holds a place on the College of Adelaide, obtained a Submit Graduate Analysis Award from the Australian institute of Nuclear Science and Engineering (AINSE).
Sources
-
G. Liang, E. Olsson, J. Zou, Z. Wu, J. Li, C.-Z. Lu, A. M. D’Angelo, B. Johannessen, L. Thomsen, B. Cowie, V. Ok. Peterson, Q. Cai, W. Ok. Pang, Z. Guo (2022) “Introducing 4s–2p Orbital Hybridization to Stabilize Spinel Oxide Cathodes for Lithium-Ion Batteries” Angew. Chem. Int. Ed. doi: 10.1002/anie.202201969